IMFS
- takes a lot of energy to break the stronger bonds
- Intermolecular forces control physical properties (ie: boiling points, melting points, viscosities)
- London Dispersion forces, Dipole-dipole forces and Hydrogen bonding are all intermolecular forces
- Strength of forces: London dispersion (weakest) → dipole-dipole → hydrogen bonds (strongest)
- Strength of the forces increase with increasing polarity
- The stronger the polarity, the higher the boiling point
- takes a lot of energy to break the stronger bonds
- Solids have the most intermolecular forces and least amount of kinetic energy
- Liquid have some intermolecular forces
- Gases have the least intermolecular forces and most amount of kinetic energy
- Heating curve equation: q= mc▲T or q=m▲H
- q= heat
- m=mass
- c=specific heat capacity
- ▲T= change in temperature
- ▲H= fusion or vaporization
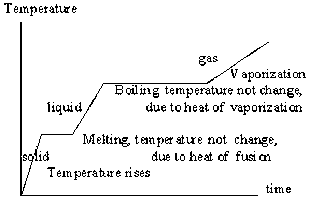
Heating Curve
- Diagonal lines are change in temperature (use q=mc▲T)
- Straight lines are phase changes, temperature does not change (use q=m▲H)
- Going up the chart from solid → gas is endothermic (absorbing energy, sign is negative [-])
- Going down the chart from gas → solid is exothermic (releasing energy, sign is positive [+])
- ▲H of fusion is phase change of solid → liquid or liquid → solid
- ▲H of vaporization is phase change of liquid → gas or gas → liquid
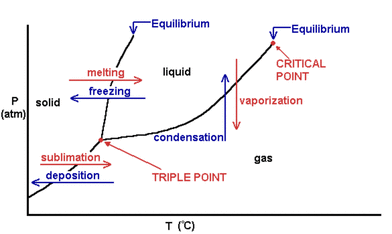
Phase Diagram
- At different pressures and temperatures, a substance is in a different state.
- The lines that border the certain state are the various points of phase changes (ie: boiling point, melting point, freezing point)
- The triple point is when all states (solid, liquid, gas) exist at the same time at equilibrium
- The critical point is when liquid and vapor can not be identified, they are indistinguishable
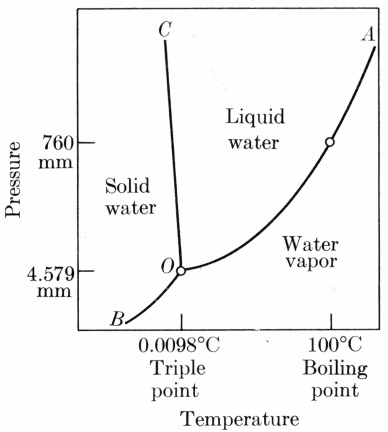
Phase Diagram of Water
Phase Diagram for Water. Digital image. Geology 202. Earth.northwestern.edu, n.d. Web. 28 May 2014.
- notice how the boundaries of the different phases differ from the image above
Phase Diagram for Water. Digital image. Geology 202. Earth.northwestern.edu, n.d. Web. 28 May 2014.
SOLUTIONS
- Negative end of water molecule, O^2-, orients itself toward the positive end of NaCl molecule, Na+
- IMFs occur between solutes and solvents
- If NaCl is added to water
- Negative end of water molecule, O^2-, orients itself toward the positive end of NaCl molecule, Na+
- molecules separate (ie: H2O) to create spaces in solvent for atoms (ie: Na+ and Cl-)
- As a solid solute dissolves in the solvent, the concentration of the solute increases along with chances of colliding with the solid
- A solution can be saturated, unsaturated or supersaturated
- Mixing, or moving to a more random state, determines solubility
- Forces between gas and solvent molecules are London Dispersion forces -they increase with increasing size and mass of gas molecules
- The greater the forces between solute and solvent, greater the solubility
- Polar dissolves in Polar
- Nonpolar dissolves in Nonpolar
- Solubility of gas in any solvent increases as pressure over solvent increases
- At equilibrium, rate at gas molecules enter solution = rate at solute molecules are released from solution
- Solid solutes solubility in water increases as temperature of solution increases
- Gas solubility in water decreases as temperature increases
